gor (1-450, His-tag) Escherichia coli Protein
Specifications
| Product Data | |
| Species | Escherichia coli |
| Expression Host | E. coli |
| Expression cDNA Clone or AA Sequence |
MGSSHHHHHH SSGLVPRGSH MGSMTKHYDY IAIGGGSGGI ASINRAAMYG QKCALIEAKE LGGTCVNVGC VPKKVMWHAA QIREAIHMYG PDYGFDTTIN KFNWETLIAS RTAYIDRIHT SYENVLGKNN VDVIKGFARF VDAKTLEVNG ETITADHILI ATGGRPSHPD IPGVEYGIDS DGFFALPALP ERVAVVGAGY IAVELAGVIN GLGAKTHLFV RKHAPLRSFD PMISETLVEV MNAEGPQLHT NAIPKAVVKN TDGSLTLELE DGRSETVDCL IWAIGREPAN DNINLEAAGV KTNEKGYIVV DKYQNTNIEG IYAVGDNTGA VELTPVAVAA GRRLSERLFN NKPDEHLDYS NIPTVVFSHP PIGTVGLTEP QAREQYGDDQ VKVYKSSFTA MYTAVTTHRQ PCRMKLVCVG SEEKIVGIHG IGFGMDEMLQ GFAVALKMGA TKKDFDNTVA IHPTAAEEFV TMR
|
| Tag | His-tag |
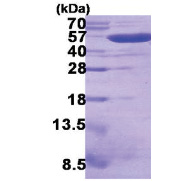
| Predicted MW | 51.2 kDa |
| Concentration | lot specific |
| Purity | >90% by SDS - PAGE |
| Presentation | Purified |
| Buffer | Presentation State: Purified State: Liquid purified protein Buffer System: 20 mM Tris-HCl buffer (pH 8.0) containing 10% glycerol, 0.1M NaCl, 1 mM DTT |
| Bioactivity | Specific: > 52 unit/ml. One unit will reduce 1.0 umol of oxidized glutathione per minute at pH 7.5 at 25°C. |
| Preparation | Liquid purified protein |
| Applications | Protocol: 1. Prepare a 1,450 ul assay buffer. The final concentrations are 75mM Potassium phosphate (pH 7.5), 2.6mM EDTA, 1mM glutathione, 0.09mM beta-NADPH, and 0.13% BSA. 2. Add 50 ul of recombinant gor protein with 0.037 ug, 0.075 ug and 0.15 ug in assay buffer 3. Mix and load 200 ul of reaction mix in to a plate well. 4. Record the decrease in A340nm for 5 minutes at 25°C. |
| Protein Description | Recombinant E. coli gor protein, fused to His-tag at N-terminus, was expressed in E.coli and purified by using conventional chromatography techniques. |
| Storage | Store undiluted at 2-8°C for one week or (in aliquots) at -20°C to -80°C for longer. Avoid repeated freezing and thawing. |
| Stability | Shelf life: one year from despatch. |
| Reference Data | |
| Summary | gor, also known as Glutathione reductase, belongs to the class-I pyridine nucleotide disulfide oxidoreductase family. The main function of the protein is to maintain high levels of reduced glutathione in the cytosol. With the concomitant oxidation of NADPH, Glutathione reductase transforms oxidized glutathione to the reduced form. The active site of the protein is a redox-active disulfide bond. |
Documents
| FAQs |
| SDS |
Resources
Recombinant Protein Resources |
{0} Product Review(s)
0 Product Review(s)
Submit review
Be the first one to submit a review
Product Citations
*Delivery time may vary from web posted schedule. Occasional delays may occur due to unforeseen
complexities in the preparation of your product. International customers may expect an additional 1-2 weeks
in shipping.






























































































































































































































































 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China